 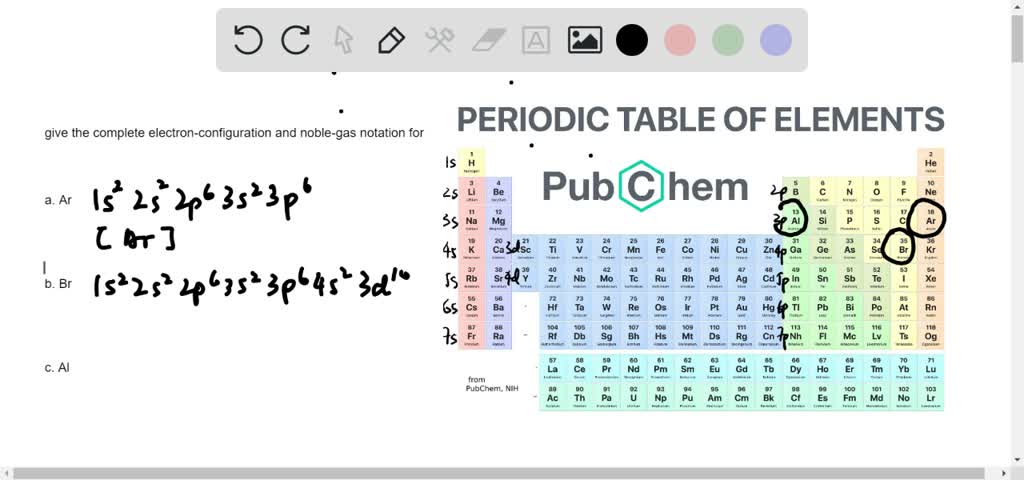
Mårtensson, "Core-Level Binding Energies in Metals," J. A sublevel which is exactly half filled is more stable than a partially filled sublevel which is not half full. Completely filled sublevels are more stable than partially filled sublevels. The electron configuration of chromium can be expressed as Ar4s. The actual electron configurations are: Cr Ar 4s1 3d5 Cu Ar 4s1 3d10 To understand why this occurs, it is important to realize that. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. (a) Explain why the relative atomic mass of cobalt is greater than the relative atomic. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Step 2: Identify the electron of interest, and ignore all electrons in higher groups (to the right in the list from Step 1). Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. Step 1: Write the electron configuration of the atom in the following form: (1s) (2s, 2p) (3s, 3p) (3d) (4s, 4p) (4d) (4f) (5s, 5p). To write the configuration for the Chromium ions, first we need to write the electron configuration for just Chromium (Cr). They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). Crs electron configuration, following the model would be: (1s2 2s2 2p6 3s2 3p6 4s23d40, but instead it is (1s2 2s2 2p6 3s2 3p6 4s13d5. The data are adapted from references 1-3. Ground state electron configurations are the foundation for understanding molecular bonding, properties, and structures. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The cobalt atom has a radius of 125 pm and a Van der Waals radius of 192 pm. The number of electrons in each of cobalt's shells is 2, 8, 15, 2 and its electron configuration is Ar3d 7 4s 2. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. Cobalt (atomic symbol: Co, atomic number: 27) is a Block D, Group 9, Period 4 element with an atomic weight of 58.933195. The electron configuration of a neutral cobalt atom is written as follows. 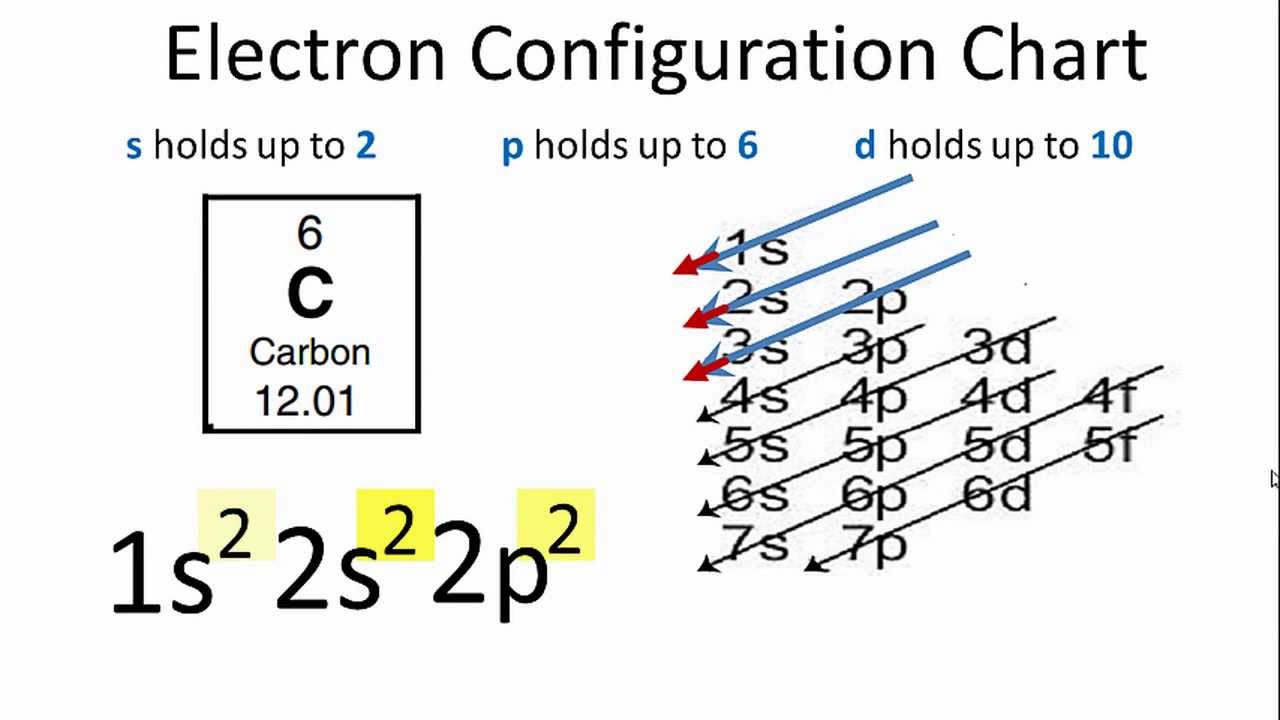
Example: Lets consider the chemistry of cobalt which forms complexes that contain either Co 2+ or Co 3+ ions. The relationship between the electron configurations of transition-metal elements and their ions is complex. 
All values of electron binding energies are given in eV. The Electron Configuration of Transition-Metal Ions. 1967, 47, 1300.Įlectron binding energies Electron binding energies for cobalt. These effective nuclear charges, Z eff, are adapted from the following references: 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
